Understanding the temporary pause on J&J vaccines as the variants spread
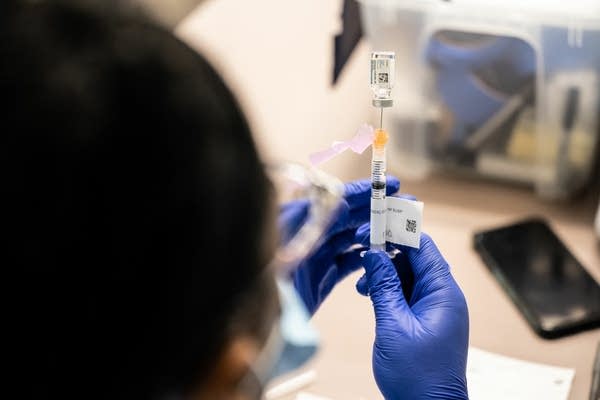
Nurse Jasmine Cowherd fills a syringe with a dose of the Johnson & Johnson COVID-19 vaccine at Wayside Christian Mission on March 15 in Louisville, Ky.
Jon Cherry | Getty Images file
Go Deeper.
Create an account or log in to save stories.
Like this?
Thanks for liking this story! We have added it to a list of your favorite stories.



